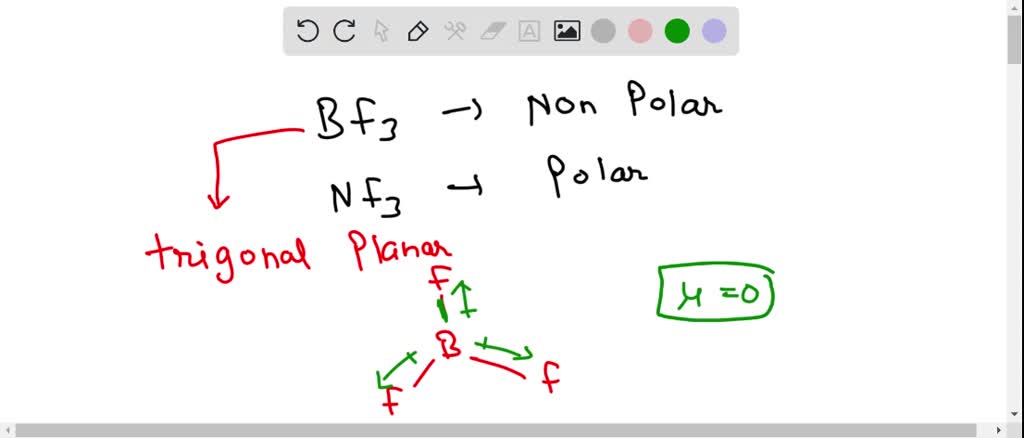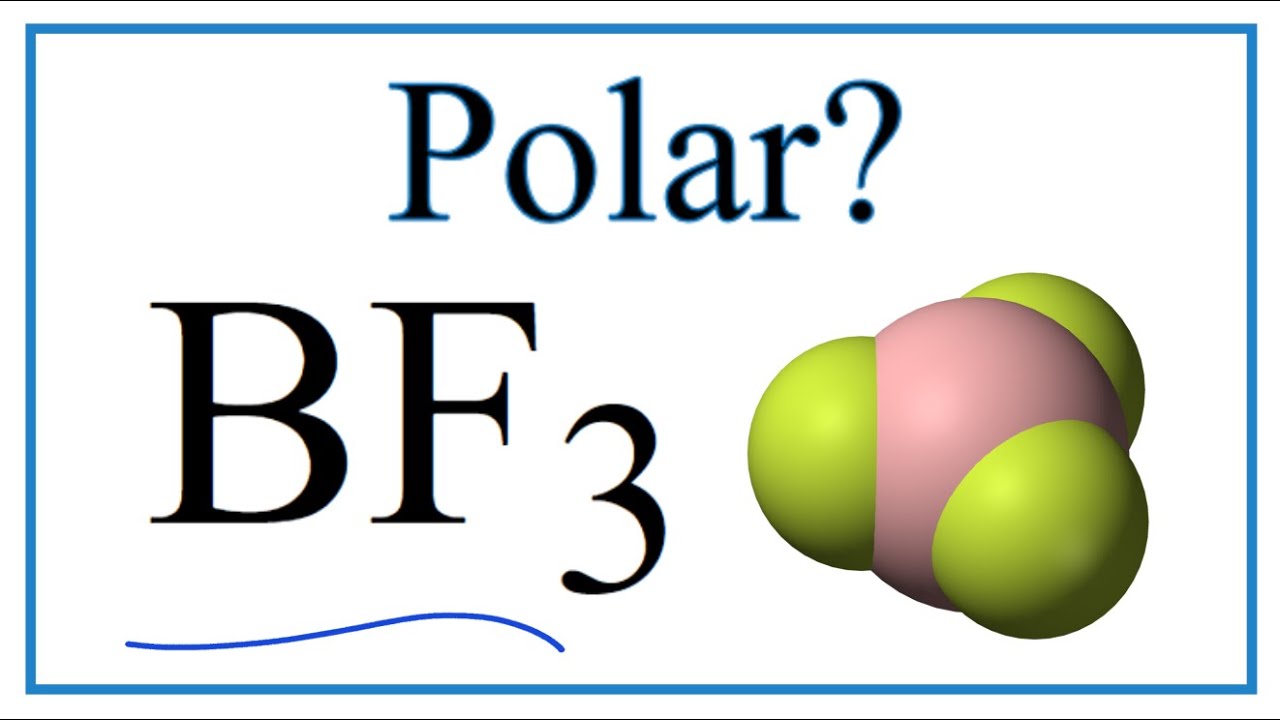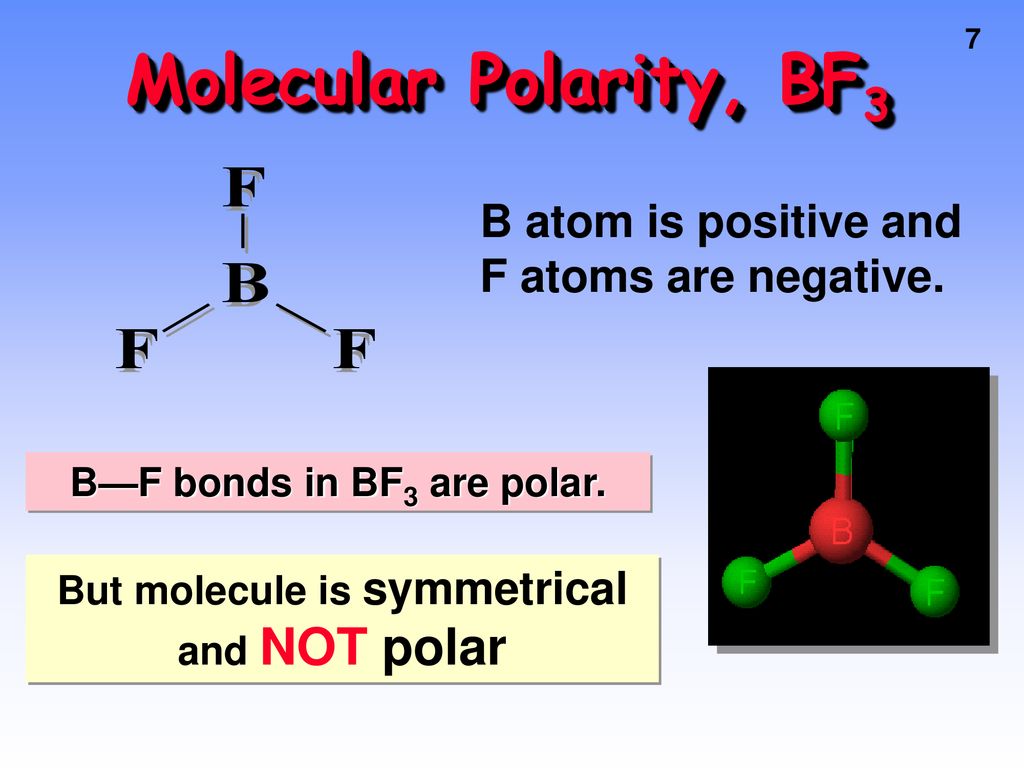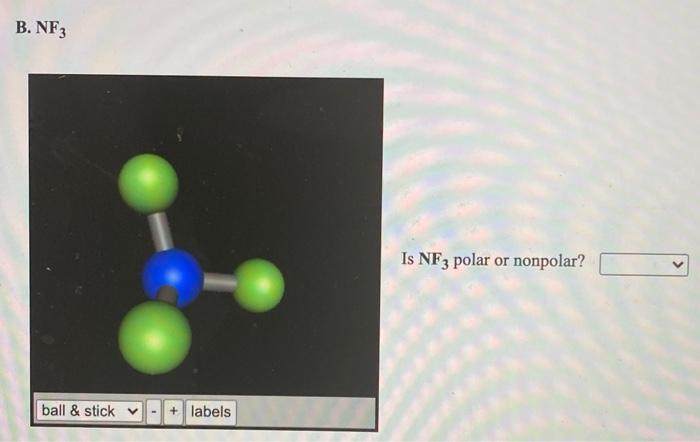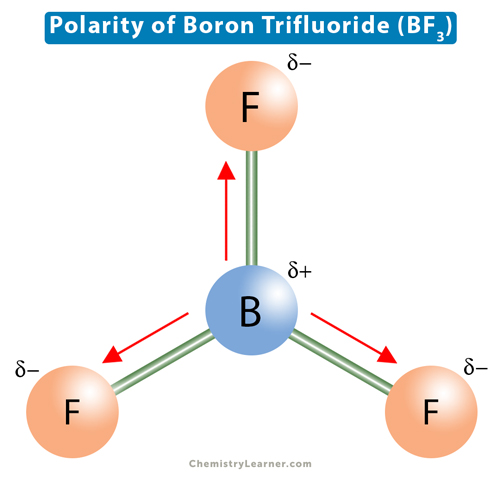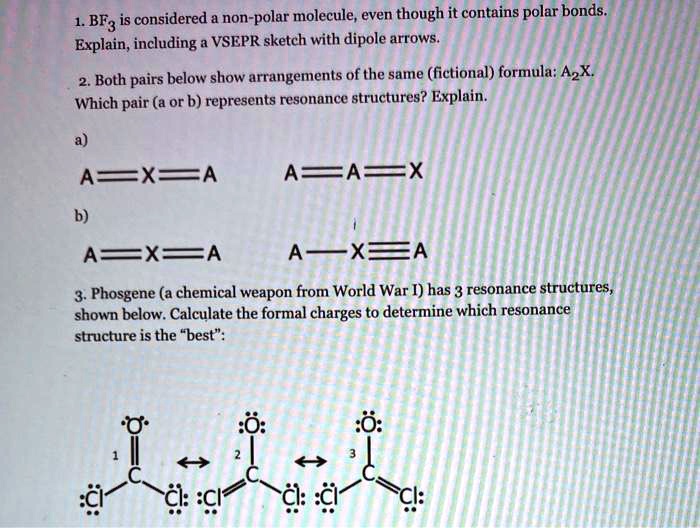
SOLVED: BF3 is considered a nonpolar molecule, even though it contains polar bonds. Explain, including a VSEPR sketch with dipole arrows. Both pairs below show arrangements of the same (fictional) formula: AzX.
Boron trifluoride (displaystyle B{ F }_{ 3 }) is a nonpolar molecule, whereas ammonia (displaystyle N{ H }_{ 3 }) is a polar molecule. The difference in polarities is related to the
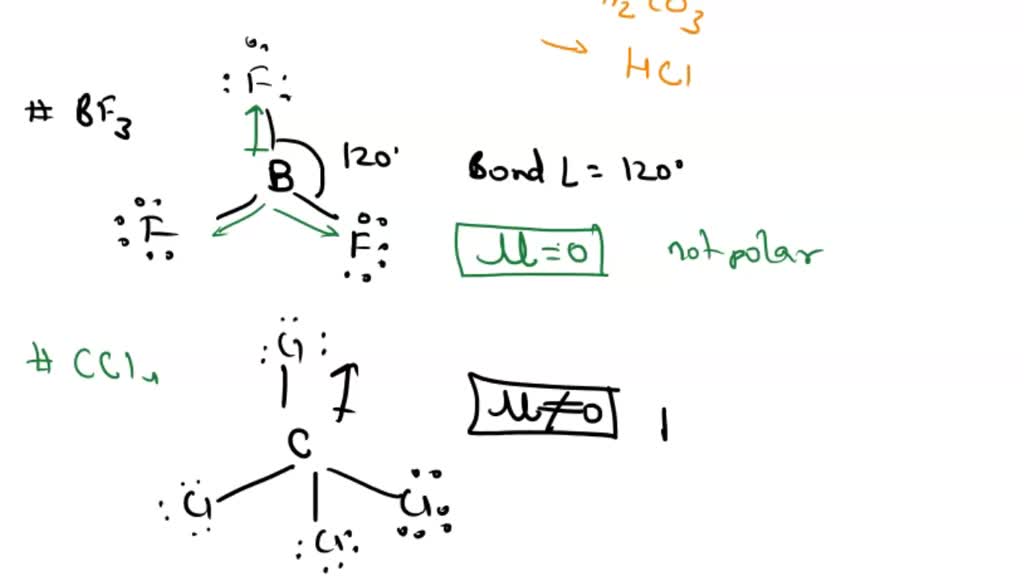
SOLVED: Determine the bond polarity, molecular polarity; electron geometry molecular geometry and bond angles and Lewis structure: 1 BF3 2 CCI 3 PCI; HCO 5 HCI

BF3 Molecular Geometry, Shape and Bond Angles (Boron Trifluoride) | BF3 Molecular Geometry, Shape and Bond Angles (Boron Trifluoride) Today in this video, we help you determine the molecular geometry of a